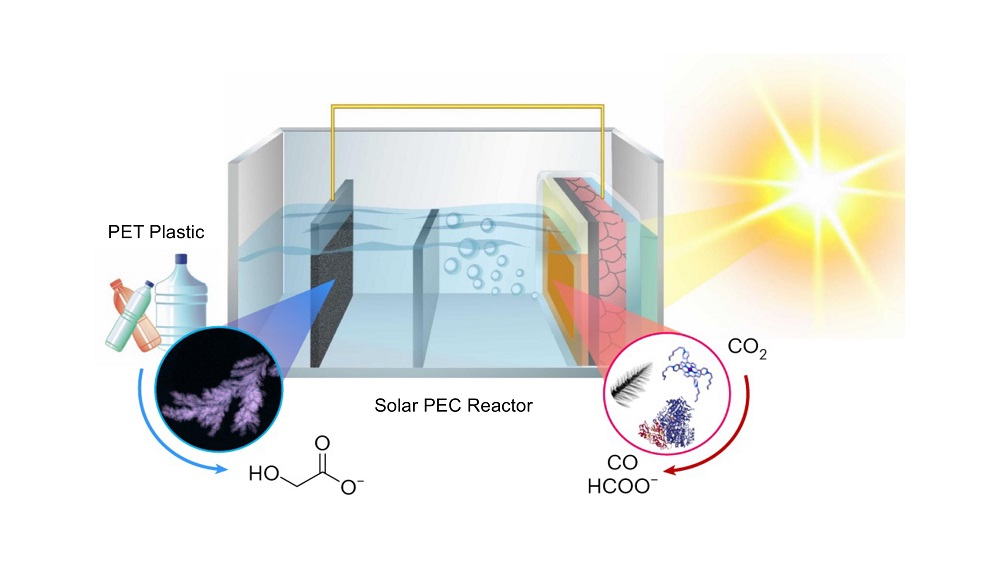
A solar powered reactor has been developed by scientists at the University of Cambridge which can convert plastics and greenhouse gases into useful products and sustainable fuels.

The reactor converts the carbon dioxide (CO2) and plastics into different products that are useful in a range of industries. In tests, CO2 was converted into syngas, a key building block for sustainable liquid fuels, and plastic bottles were converted into glycolic acid, which is widely used in the cosmetics industry. The system can easily be tuned to produce different products by changing the type of catalyst used in the reactor.
Professor Erwin Reisner from the Yusuf Hamied Department of Chemistry, explained:
“Converting waste into something useful using solar energy is a major goal of our research. Plastic pollution is a huge problem worldwide, and often, many of the plastics we throw into recycling bins are incinerated or end up in landfill.
“Developing a circular economy, where we make useful things from waste instead of throwing it into landfill, is vital if we’re going to meaningfully address the climate crisis and protect the natural world.
“And powering these solutions using the Sun means that we’re doing it cleanly and sustainably.”

The solar reactor has two separate compartments: one for plastic, and one for greenhouse gases. It uses a light absorber based on perovskite – a promising alternative to silicon for next-generation solar cells.
Tests of the reactor under normal temperature and pressure conditions showed that the reactor could efficiently convert PET plastic bottles and CO2 into different carbon-based fuels such as CO, syngas or formate, in addition to glycolic acid. The Cambridge-developed reactor produced these products at a rate that is also much higher than conventional photocatalytic CO2 reduction processes.
The research was supported in part by the European Union, the European Research Council, the Cambridge Trust, Hermann and Marianne Straniak Stiftung, and the Engineering and Physical Sciences Research Council (EPSRC), part of UK Research and Innovation (UKRI). Erwin Reisner is a Fellow of St John’s College, Cambridge. The team will now develop the reactor to produce more complex molecules.
Click on this link to access the full report: Photoelectrochemical CO2-to-fuel conversion with simultaneous plastic reforming via Nature Synthesis




Leave a Reply